Modular Access to meta-Substituted Benzenes via Mo-Catalyzed Intermolecular Deoxygenative Benzene Formation
Authors: Yi-Zhe Yu, Jin Bai, Jia-Min Peng, Jia-Sheng Yao, and Chun-Xiang Zhuo*
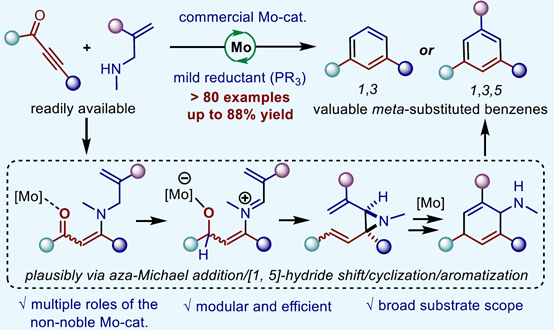
Abstract: The substituted benzene derivatives are essential to organic synthesis, medicinal chemistry, and material science. However, the 1,3-di- and 1,3,5-trisubstituted benzenes are far less prevalent in small-molecule drugs than other substitution patterns, likely due to the lack of robust, efficient, and convenient synthetic methods. Here, we report a Mo-catalyzed intermolecular deoxygenative benzene-forming reaction of readily available ynones and allylic amines. A wide range of unsymmetric and unfunctionalized 1,3-di- and 1,3,5-trisubstituted benzenes were obtained in up to 88% yield by using a commercially available molybdenum catalyst. The synthetic potential of the method was further illustrated by synthetic transformations, a scale-up synthesis, and derivatization of bioactive molecules. Preliminary mechanistic studies suggested that this benzene-forming process might proceed through a Mo-catalyzed aza-Michael addition/[1,5]-hydride shift/cyclization/aromatization cascade. This strategy not only provided a facile, robust, and modular approach to various meta-substituted benzene derivatives but also demonstrated the potential of molybdenum catalysis in the challenging intermolecular deoxygenative cross-coupling reactions.
Link: https://pubs.acs.org/doi/10.1021/jacs.3c01330